Fantastic Info About How To Draw Lewis Diagrams
Check your understanding of lewis diagrams in this set of free practice questions designed for ap chemistry students.
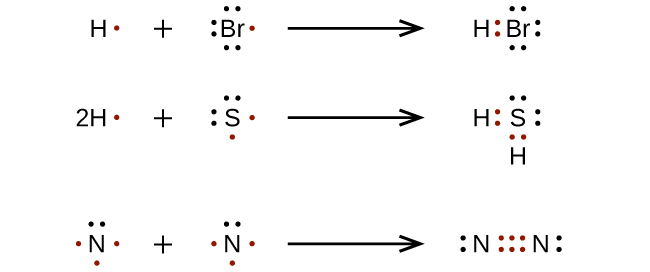
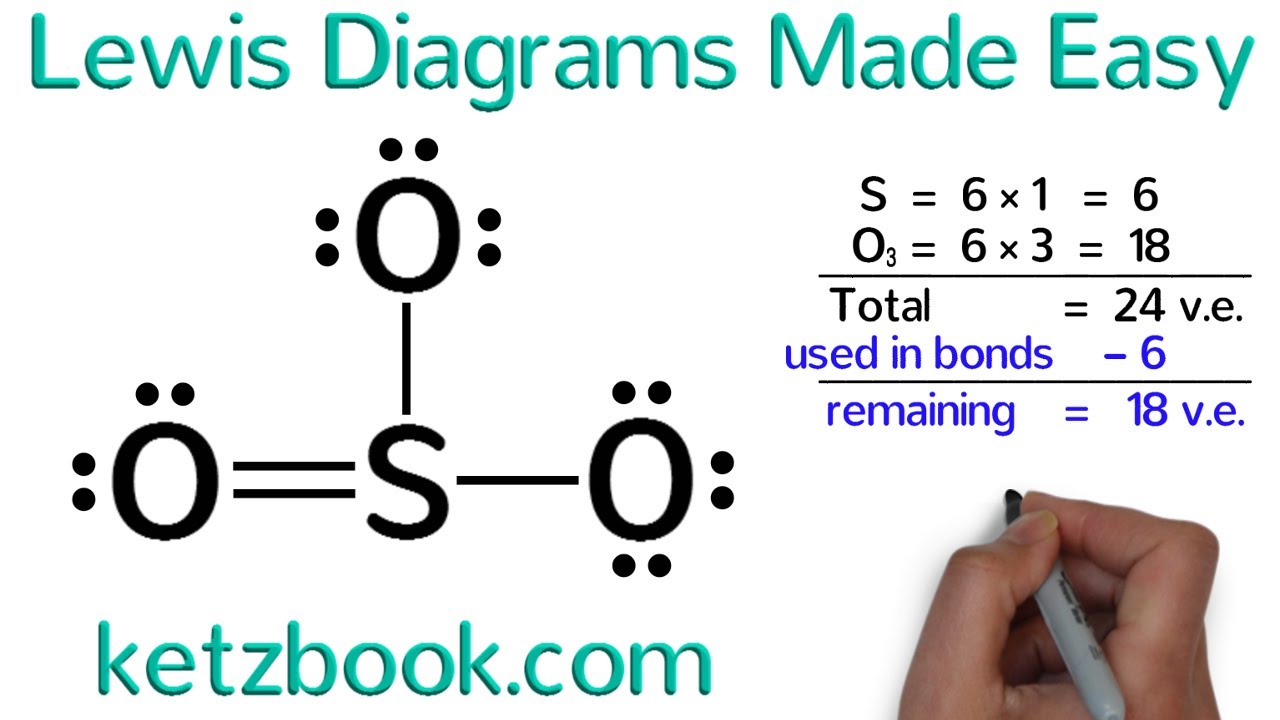
How to draw lewis diagrams. 1 chemical formula total number of valence electrons lewis dot. Place electron pairs around each. They also display the total number of lone pairs present.
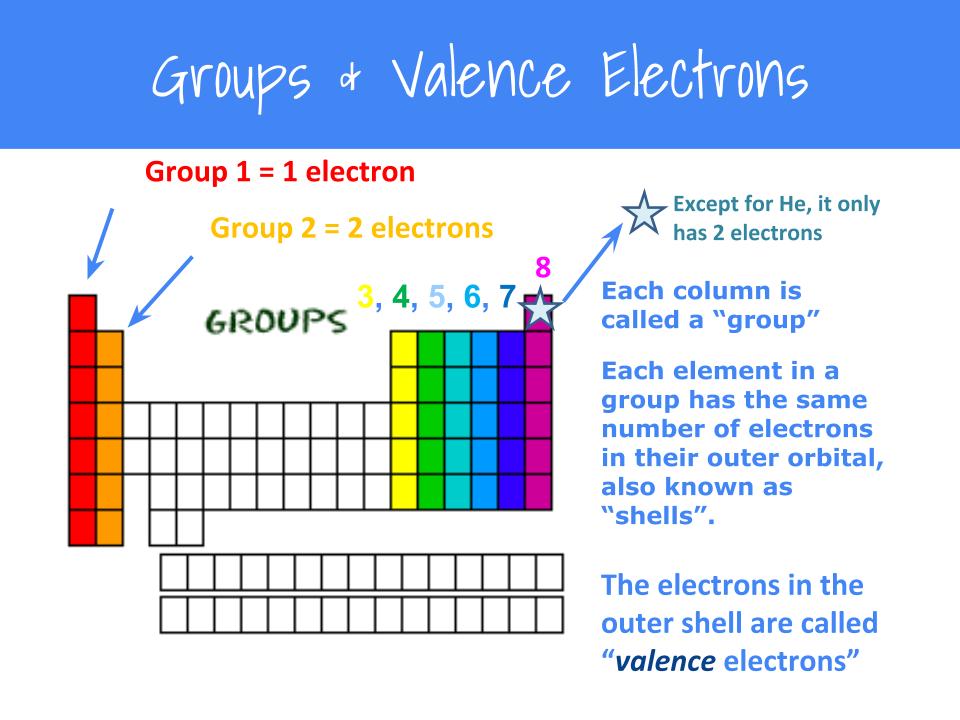
Lewis dot structures also called electron dot structures are diagrams that describe the chemical bonding between atoms in a molecule. Thus, a pair of dots represent the bond between the draw lewis dot diagrams for the following molecules: We can do this by looking at the periodic table.
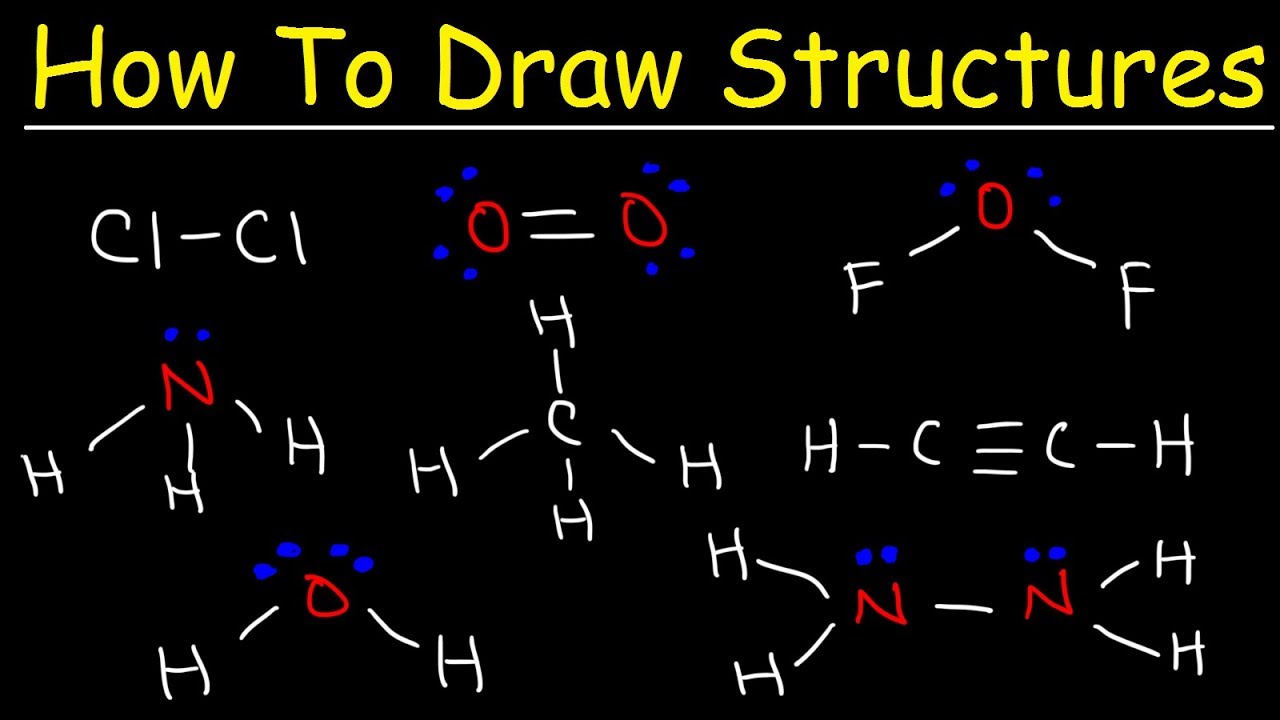
Determine the total number of valence electrons in a. When constructing a lewis diagram, keep in mind the octet rule,. Draw a lewis dot diagram for ammonia, nh 3.
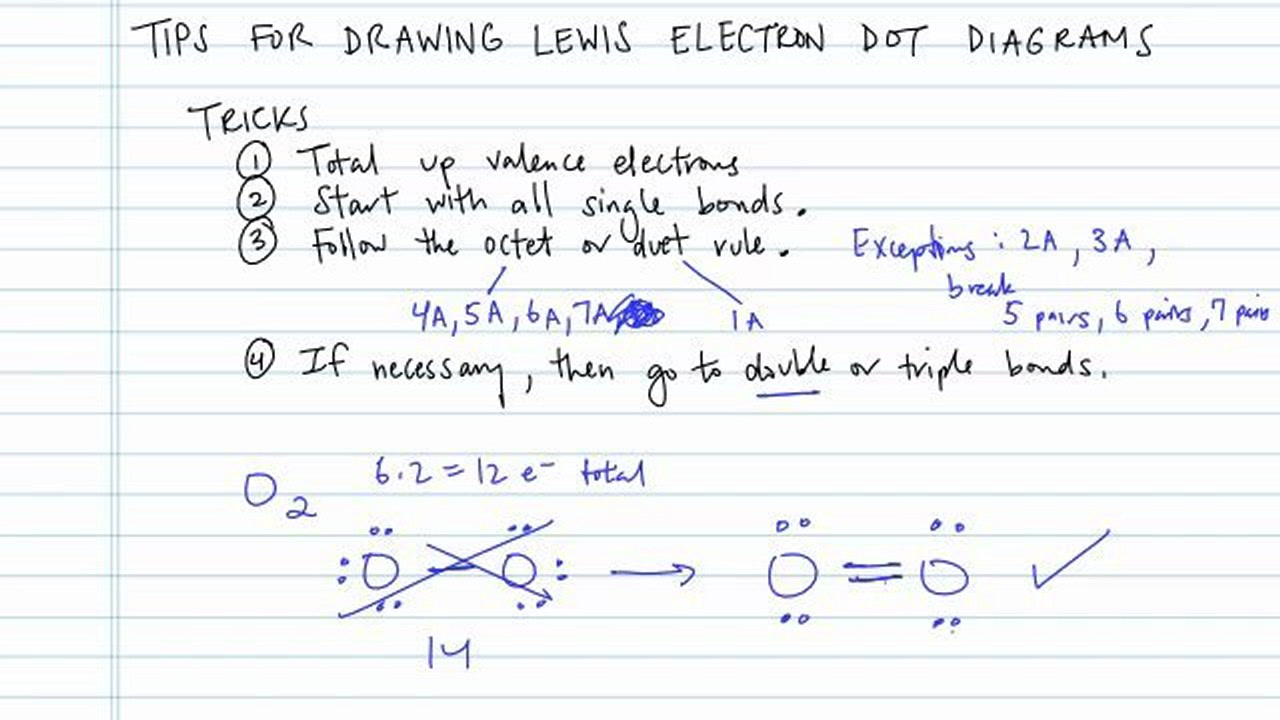
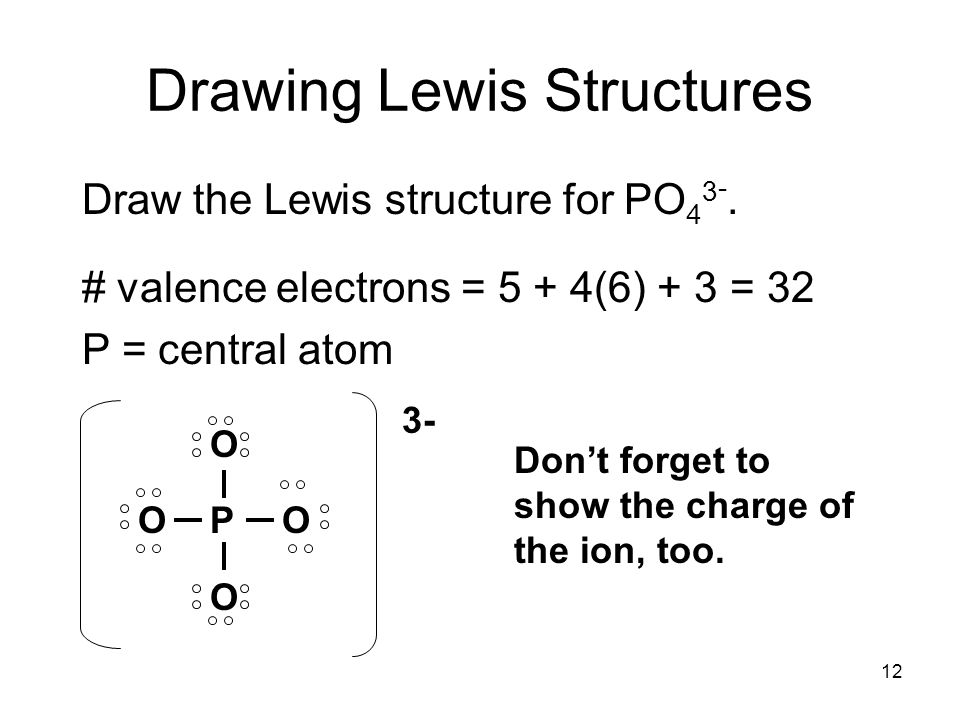
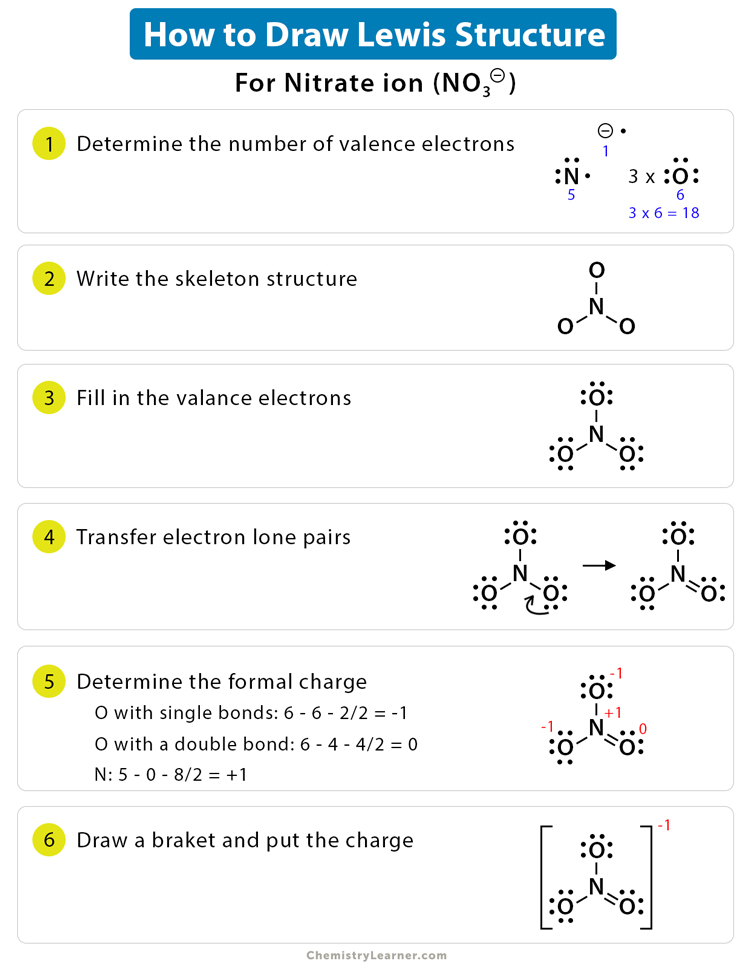
Draw skeletal structure of compound showing what atoms are bonded to each other. Add your bonds to the drawing. Steps to draw lewis structure the steps to draw the lewis structures of various types of compounds are given below:
Find the total valence electrons for the molecule. Lewis diagrams (aka lewis structures, lewis dot structures, lewis dot diagrams) are useful. I go over two lewis structure problems in this video, one problem on the easier side so that you can really learn the fundamental rules of drawing lewis stru.
First of all, we need to work out the number of valence electrons in this molecule, ammonia. This is a videoscribe tutorial of how to draw lewis diagrams for elements and simple molecules. How to draw lewis dot structures.
For a single bond, you would simply draw 1 line from the first atom to the second. (put least electronegative element in the center. Andersen shows you how to draw lewis dot diagrams for atoms and simple molecules.intro music atributiontitle:
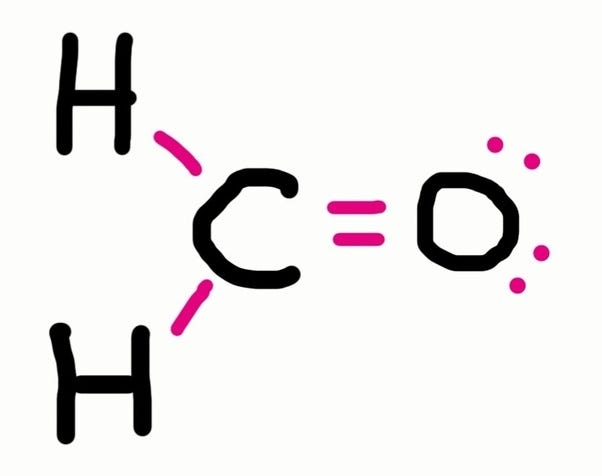
Lewis structure of oxygen \ (\left ( { { {\rm {o}}_ {\rm. Place one electron pair between each pair of adjacent atoms (as determined from the framework found in step 2) to form a single bond. Put the least electronegative atom in the center.




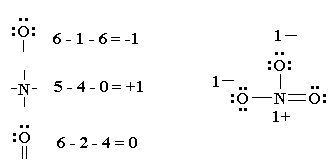

/Lewis-dot-structure-58e5390f3df78c5162b4c3db.jpg)